Page snapshot: Introduction to the domestication of maize from teosinte, including evidence from plant structure, genetics, and archaeology.
Topics covered on this page: Introduction; What is teosinte?; Morphology of teosinte; Wild relatives of maize; Balsas teosinte; Mexican teosinte; Process of maize domestication; Archaeological evidence for domestication; Microremains; Macroremains; Huaca Prieta and Paredones (Peru); Guilá Naquitz Cave (Oaxaca, Mexico); Coxcatlán and San Marcos caves (Puebla, Mexico); Ocampo Caves (Tamaulipas, Mexico); El Gigante rockshelter (Honduras); Southwestern U.S.A.; Maize becomes a staple crop; Resources.
Credits: Funded by the National Science Foundation. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation. Some text on this page comes from a revised manuscript of the Teacher-Friendly Guide to the Evolution Maize (Carlyn Buckler, Dhyan Palanichamy, and Andrielle Swaby, 2019). Additional text and revisions by Elizabeth J. Hermsen (2022-2023).
Updates: Page last updated May 2, 2023.
Image above: Teosinte cultivated at the ethnobotanical museum in Oaxaca, Mexico. Photo by Barnardo Bolaños (Wikimedia Commons, Creative Commons Attribution-ShareAlike 3.0 Unported license, image cropped and resized).
Introduction
Maize in the form that we are accustomed to today has only existed for approximately 6000 years. Maize is derived from an earlier ancestral plant called teosinte, which was first domesticated in Mexico. The ears of teosinte do not look much like the ears of modern maize, and the reasons that ancient Mesoamericans first adopted teosinte as a crop are not well understood. This page explores teosinte, the changes that teosinte underwent during its domestication into maize, and some of the archaeological evidence behind the domestication of maize.
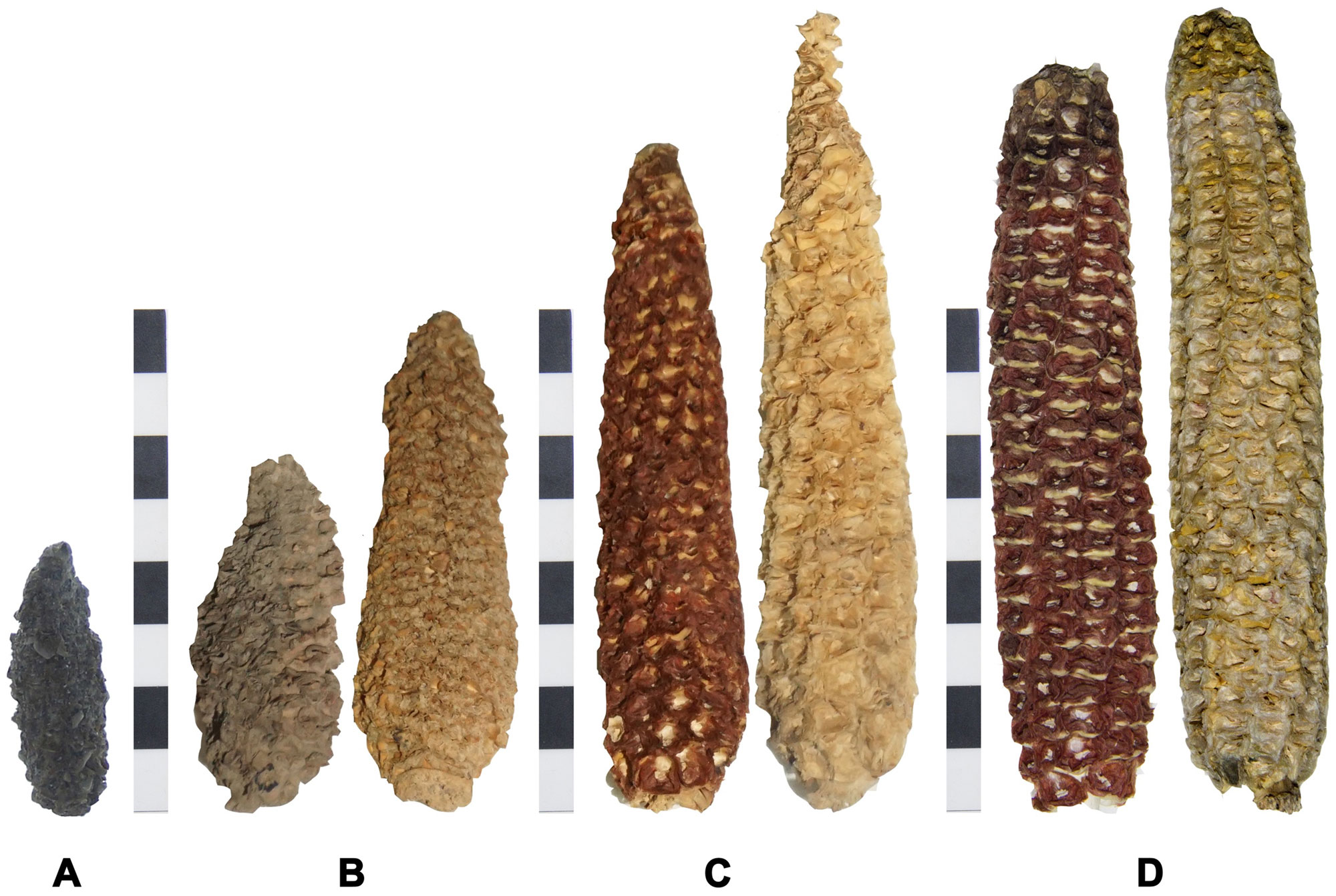
Ancient and modern cobs of maize from the Tarapacá region of northern Chile. The cobs come from the following sites and are estimated to be the following ages: A. Pircas, 2320 to 1420 years old. B. Tarapacá 13, 930 to 490 years old. C. Tarapacá Viejo, 662 to 350 years old. D. Camiña (left) and Tarapacá (right), modern. Scale bars = 8 centimeters (about 3.15 inches). Source: Figure 2 from Elgueta et al. (2019) PLoS ONE 14(1): e0210369 (Creative Commons Attribution 4.0 International license, image resized).
What is teosinte?
Teosinte is the name for a group of a group of wild grasses in the genus Zea that are native to Mexico and Central America. Teosintes include the species Zea diploperennis, Z. luxurians, Z. nicaraguensis, and Z. perennis. The species Zea mays itself also includes three subspecies of teosinte: Z. mays subsp. huehuetenangensis, Z. mays subsp. mexicana, and Z. mays subsp. parviglumis.
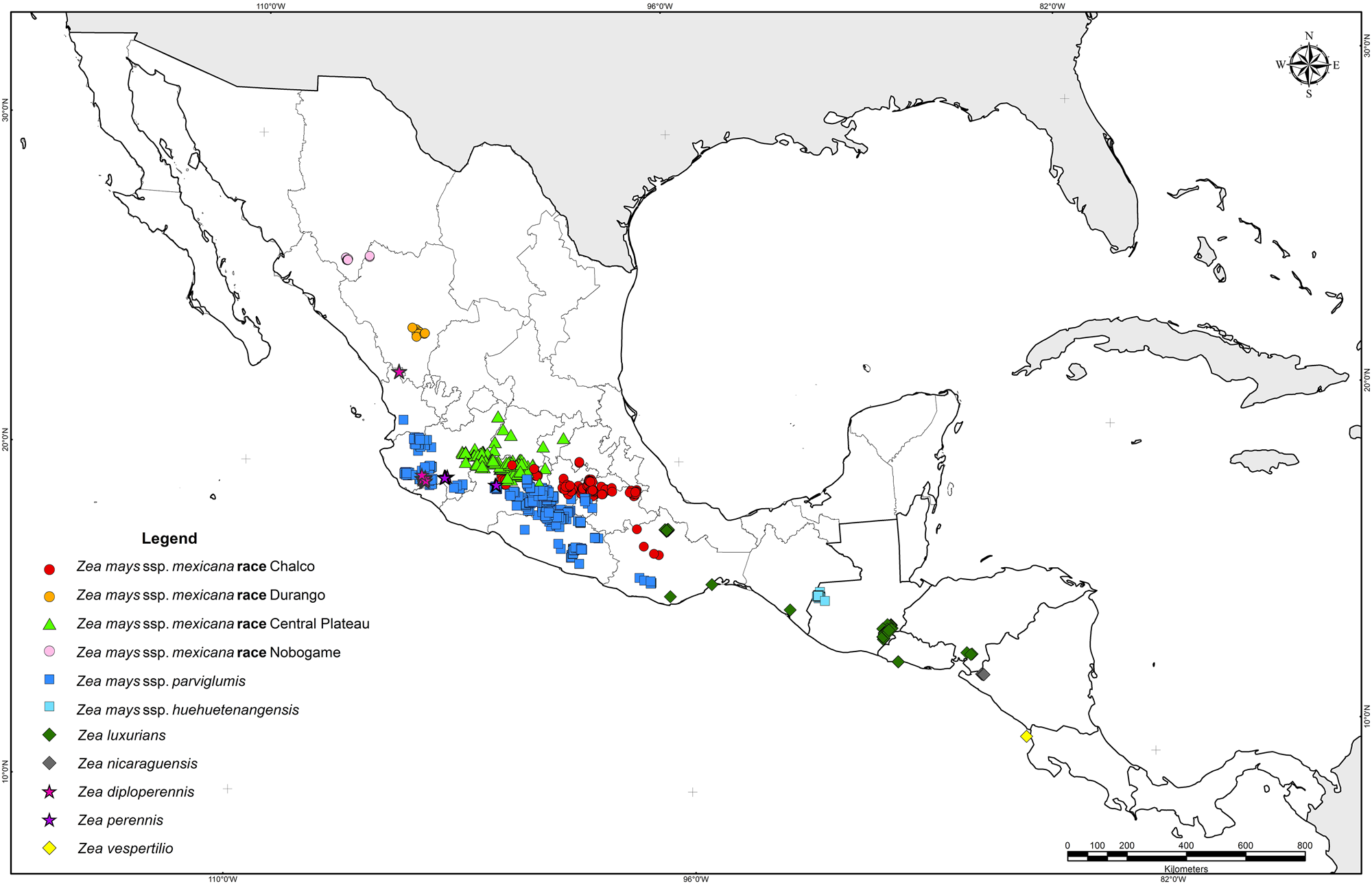
Locations of observations and collections of wild teosintes in Mexico and Central America. Source: Figure 1 from Sánchez González et al. (2018) PLoS ONE 13(2): e0192676 (Creative Commons0 1.0 Universal/Public Domain Dedication).
Morphology of teosinte
Teosinte is a tall, branching grass. Like maize (corn), it is monoecious. This means that each plant produces separate male (staminate or pollen-producing) and female (carpellate or ovule-producing) florets (tiny flowers). The male and female florets are in separate inflorescences (groups of flowers).
The male inflorescence, or tassel, extends vertically from the end of each major branch of the teosinte plant. Each tassel consists of a thin central branch with many delicate lateral (side) branches (also called rames). The lateral branches of the tassel bear tiny spikelets, each with two florets. Each floret has three stamens, or pollen-producing structures.

Teosinte plants. Left: Zea diploperennis. Right: Zea mays subspecies mexicana. Photo credits: Zea diploperennis by Jeffdelonge (Wikimedia Commons, Creative Commons Attribution-ShareAlike Unported license, image cropped and resized); Zea mays subspecies mexicana (Wikimedia Commons, via USDA PLANTS, public domain).

Herbarium sheets (sheets with dried, pressed plants attached) showing tassels (male inflorescences or staminate inflorescences) of teosinte. Left: Zea diploperennis. Right: Zea nicaraguensis. Photo credits: Left photo (NY 431763) and right photo (NY 468401) by the New York Botanical Garden on GBIF.org (Creative Commons Attribution 4.0 International license, images cropped and resized).
The female inflorescences, called ears, rames, or spikes, occur at the ends of short side branches of the teosinte plant. The branches bearing the ears are found in the leaf axils (a leaf axil is the upper angle where the leaf attaches to the stem). Each ear is enclosed by a thin husk made up of modified leaves.
Each ear has 5 to 15 spikelets alternating in two vertical rows (in other words, the spikelets are in a distichous arrangement, as is common in grasses). Each individual spikelet consists of a floret surrounded by hard glumes. A silk (a long, feathery stigma) extends from each spikelet.
In some cases, both male and female florets occur in the same inflorescence at the tip of a stem. In these inflorescences, the female florets are near the base and the male florets are near the tip.

Perennial teosinte (Zea diploperennis). Left: Ears (female inflorescences) on a stem. Right: Detail of an inflorescence with with both female spikelets and male spikelets. Photo credits: Left photo and right photo by Matt Lavin (Wikimedia Commons, Creative Commons Attribution-ShareAlike 2.0 Generic license, images cropped, resized, and labeled).
The mature female spikelets are fruits called grains or kernels. Each kernel is sealed within a stony casing called a fruitcase. The fruitcase is made up of the one of the hard glumes of the spikelet and a piece of the axis bearing the spikelet. When the teosinte ear is mature, the kernels disarticulate (break apart) from one another and fall to the ground still wrapped in their protective fruitcases.

Teosinte kernels. Photo by Michael Chauvet (Wikimedia Commons, Creative Commons Attribution-ShareAlike 3.0 Unported license, image resized).
Wild relatives of maize
Balsas teosinte (Zea mays subspecies parviglumis)
Genetic and archaeological evidence together suggest that teosinte was the ancestor of modern maize, and that its domestication began around 9000 years ago in the Balsas River region of Guerrero and Michoacán, Mexico. While most species of teosinte are distinct from maize both genetically, Balsas teosinte (Zea mays subspecies parviglumis) shares a close genetic relationship with domesticated maize (Zea mays subspecies mays) and is considered to be its direct ancestor. Balsas teosinte grows wild in moist habitats along streams and hillsides in the Central Balsas River Valley, around elevations of 1312 to 5906 feet (400 to 1800 meters). Wild teosinte in this region also has the highest genetic diversity, suggesting that this is the site of the plant’s initial domestication, because diversity in a wild plant population provides more options and variants to select for in a crop.

Balsas teosinte (Zea mays subspecies parviglumis), showing tassels. Photo by Hugh H. Iltis (University of Wisconsin-Digitized Collections, Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International license).
Mexican teosinte (Zea mays subspecies. mexicana)
Another variety of teosinte, Mexican teosinte (Zea mays subspecies mexicana), has also contributed genetic material to modern maize. This subspecies of teosinte is closely related to Balsas teosinte. While both species live in the same general geographic region of Mexico, Mexican teosinte is adapted to drier environments and grows at higher elevations, between about 5249 to 8858 feet (1600 and 2700 meters). Genetic traits from Mexican teosinte may have helped early maize adapt to higher elevations and more arid conditions.

Mexican teosinte (Zea mays subspecies mexicana) plants. Photo by Ignacio Torres García (iNaturalist, Creative Commons Attribution-NonCommercial 4.0 International license, image resized).
Process of maize domestication
It is unclear why ancient Mesoamericans began consuming and cultivating teosinte. The wild plants probably did not yield enough energy from grain to make them major sources of food, and maize was probably a minor crop for thousands of years after cultivation began. Some scientists have speculated that teosinte may have been targeted as a food source by early Mesoamericans for reasons other than its edible grain, such as for its relatively sweet stems or for use as a vegetable. Another theory is that teosinte grains were popped like popcorn in order to release the starch and minimize processing time.
The transformation of teosinte into maize through the process of domestication altered several key characters: 1) the amount of stem branching, 2) the size of the ears and number of kernels per ear, 3) the fruitcase, and 4) the retention of kernels on the plant. Some evidence for the transformation of the teosinte into maize comes from the archaeological record. The transformations that happened during domestication can also be inferred by comparing the structures of modern maize and teosinte plants, as well as by studying the genetics of modern maize, ancient maize, and modern teosinte.
Differences between teosinte and maize
| Teosinte | Maize | |
|---|---|---|
| Branching | Plants branched, ears on all branches | Single upright stem with lateral ears |
| Size of ears | Small | Large |
| Rows of kernels per ear | 2 | 8 to 22 |
| Hard fruitcase (wall) surrounding seed | Present | Absent |
| Ears | Ears shatter (break apart) to scatter the kernels when mature | Ears do not shatter, and kernels remain attached when mature |
Teosinte plants are highly branched, whereas maize plants have a single stem and short lateral (side) branches that each end in a single ear. The robust stems of maize can support the large, relatively heavy ears that the plant produces. The unbranched structure of the plant also makes harvest of the ears easier. The switch from the highly branched growth form of teosinte to the single-stalked growth form of maize with lateral ears (ears on the side of the stem) is controlled by expression of the gene teosinte branched 1 (tb1). Genetic analyses of ancient maize cobs from Tehuacán, Mexico (discussed below), indicates that the allele (gene variant) of tb1 found in modern maize was already established in ancient maize about 5000 years ago.
The cob is one of the most striking traits of modern maize. The maize cob is much thicker than the rachis (main axis) of the teosinte ear. Maize kernels are arranged in multiple rows on the cob, rather than alternating in only two rows, as in teosinte. The differing size of the ears and arrangement of the kernels means that each maize ear has many more kernels than each teosinte ear. The earliest domesticated maize cobs were small with kernels arranged like those of teosinte. Some maize cobs that are more than 4000 years old already had 12 to 14 rows of kernels, although they were still small compared to cobs of modern maize.
Maize kernels are naked and not individually enclosed in a protective fruitcase like the kernels of teosinte. A single mutation in a single gene called teosinte glume architecture1 (tga1) may have been responsible for the switch from hard fruitcases to naked kernels during domestication. The loss of fruitcases was important, because it made the kernels of maize much easier to process.
Ears that do not shatter (break apart) are critical for harvesting maize, as non-shattering ears to not scatter their kernels. With the loss of shattering ears, maize lost the ability to self-propagate by spreading its grain, making it dependent on human propagation for its continued survival. The earliest maize cobs in the archeological record had non-shattering ears.

Ears of teosinte (top), maize (bottom), and a maize-teosinte hybrid (center). Photo by John Doebley (Wikimedia Commons, Creative Commons Attribution 3.0 Unported license).
Ears of Conico Norteño maize landrace from Mexico. Photo by CIMMYT (flickr, Creative Commons Attribution-NonCommercial-ShareAlike 2.0 Generic license, image cropped and resized).

An ear of corn in cross section (cut horizontally) to show the structure. This cob has 18 rows (9 paired rows) of kernels. Notice that the glumes are small and thin, whereas the cob is thick. Gl = glume. Photo by Picdrome Public Domain Pictures on flickr (CC0 1.0 Universal/public domain dedication).
Archaeological evidence for domestication
The process of domesticating maize may have begun as long as 9000 years ago, although the archaeological record of early maize is sparse. The lack of many maize remains in sites dating to the early history of maize may indicate that there was a significant time gap between when people in Mesoamerica first began to domesticate maize and when it was transformed into a staple food crop.
People in some regions of the Americas first began to be rely on maize as a staple crop only between about 4700 and 4000 years ago, based on the archaeological record. Up to that time, maize may have been cultivated in part as a vegetable or to provide grain for fermented beverages rather than as a grain for food (however, these are hypotheses).

Kernels of teosinte in a basket. Photo by CIMMYT (flickr, Creative Commons Attribution-NonCommercial-ShareAlike 2.0 Generic license, image cropped and resized).
Microremains
Microremains are the oldest evidence for maize in the archaeological record. Microremains of maize include pollen, phytoliths, and starch grains. While it is difficult if not impossible to date these microremains themselves due to their tiny size, researchers usually date other organic material (such as peat, charcoal, or other large plant remains) found in association with the samples. Furthermore, distinguishing microremains produced by domesticated maize from those produced by teosinte can be difficult.
The oldest maize microremains are roughly 8700 years old, and were found at the Xihuatoxtla site in the Balsas Valley of Mexico. These remains include starch grains and phytoliths. Younger maize pollen grains that are about 7100 years old were found in the sediment near an ancient lagoon in Tabasco, Mexico. The grains found in Tabasco were identified as belonging to the grass family (Poaceae), but are larger than those of most tropical grasses, meaning that they belong to either teosinte or domesticated maize. Since teosinte does not grow naturally in the Tabasco region, the pollen most likely belongs to early maize.
The oldest maize microremains from outside of Mexico are about 7800 years old and were discovered in the Cueva de los Ladrones (Robber's Cave) along Panama’s Pacific coast. These remains include pollen, phytoliths, and even maize starch grains that were attached to the grinding surfaces of stone food-processing tools found in the cave. The Aguadulce (Freshwater) site in Panama yielded maize phytoliths that are only slightly younger, at about 7750 years old. Maize microremains dating between about 6700 and 3500 years old have been identified at many other sites in the region spanning from Mexico through Central and northern South America.

Scanning electron micrographs (photos taken with a scanning electron microscope, or SEM) of modern maize pollen in two views (other types of smaller pollen grains can be seen in the background). Left photo and right photo by Louisa Howard, Dartmouth College EM Facility (Cell Image Library, public domain).
Macroremains
Very few ancient maize cobs or kernels date to more than 4000 years old. Ancient cobs are important, because scientists can study both their morphology (form, physical traits) and their genetics to reconstruct how maize changed through time. These ancient cobs demonstrate that the story of maize domestication is complicated. While maize originated in Mexico, genetic and archeological evidence suggests that maize was transformed in South America, and, later, improved types of maize spread back northward to maize's region of origin. Maize first reached the U.S. Southwest through the Mexican highlands.
Huaca Prieta and Paredones (Peru)
Some of the oldest distinctly recognizable maize cobs are from Huaca Prieta and Paredones in coastal Peru. These cobs are of a type of early popcorn, with the oldest dating to about 6800 to 6500 years ago. The 6800- to 6500-year-old cob has eight rows of kernels and is a little more than 3 centimeters (1.2 inches) long.
Interestingly, this early maize cob from Peru has more maize-like features than some younger cobs found in Mexico (see below), which are more similar in form and organization to ears of teosinte. The changes seen in the early Peruvian maize may have been caused by human selection following the spread of maize out of Mexico and into northern South America, whereas the more teosinte-like forms of maize persisted longer nearer maize's region of origin.

Huaca Prieta site, Peru, 2007. Photo by Véronique Debord-Lazaro (flickr, Creative Commons Attribution-ShareAlike 2.0 Generic license).
Guilá Naquitz Cave (Oaxaca, Mexico)
Early maize cobs have been discovered at the Guilá Naquitz cave in Oaxaca, Mexico. These cobs are about 6250 years old. The cobs are tiny—the largest is only about an inch (2.5 centimeters) long—and are clearly non-shattering. Two of the cobs had kernels arranged distichously (in two rows, with kernels alternating on either side of the cob), like teosinte. One cob had two rows of paired kernels, for a total of four rows of kernels. Modern maize cobs also have paired kernels.
Although the glumes found on the tiny ears from Guilá Naquitz did not form the hard fruitcase around the kernel that is characteristic of teosinte, they are still longer and firmer than those of modern maize. The lack of fruitcases on the Guilá Naquitz cobs indicates that while early farmers had already selected for plants with exposed kernels, this trait would continue to be modified well into the future.

Guilá Naquitz cave, Oaxaca, Mexico. Photo by The Shifted Librarian (flickr, Creative Commons Attribution-NonCommercial-ShareAlike 2.0 Generic license, image resized).
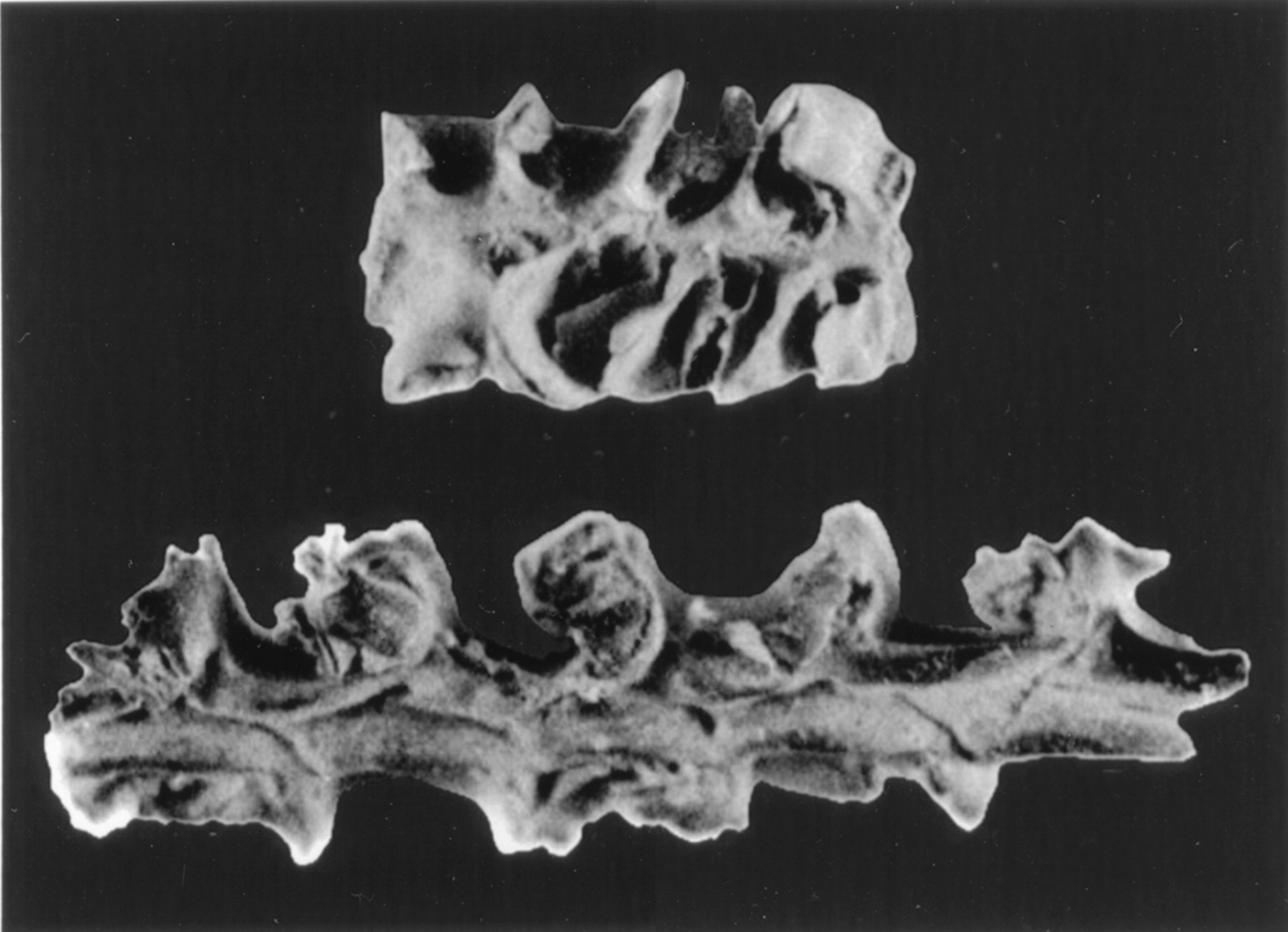
Examples of corn cobs from Guilá Naquitz cave, Oaxaca, Mexico. These cobs have been dated to about 6250 years before present. The longer cob is about 2.5 centimeters. Figure 2 from D. R. Piperno and K. V. Fannery (2001) Proceedings of the National Academy of Sciences 98: 2101–2103. Copyright 2001, The National Academy of Sciences, used following PNAS rights and permissions policy).
Coxcatlán and San Marcos caves (Puebla, Mexico)
Other important early maize remains from Mexico come from the Coxcatlán and San Marcos caves in the Tehuacán Valley, Puebla. Some of the cobs from these sites are more than 5000 years old, with the oldest dating to about 5900 years ago. The cobs from the Tehuacán caves have varying numbers of kernel rows, rarely two rows of paired kernels (four rows total), more commonly eight or ten rows of kernels.
Researchers studied the genetics of maize cobs from Tehuacán. While the cobs were more closely related to modern maize than teosinte, they had mixtures of genetic similarities to both Balsas teosinte and maize. Therefore, genetic analysis of the Tehaucán cobs showed that the process of maize domestication was still ongoing in this region about 5000 years ago.
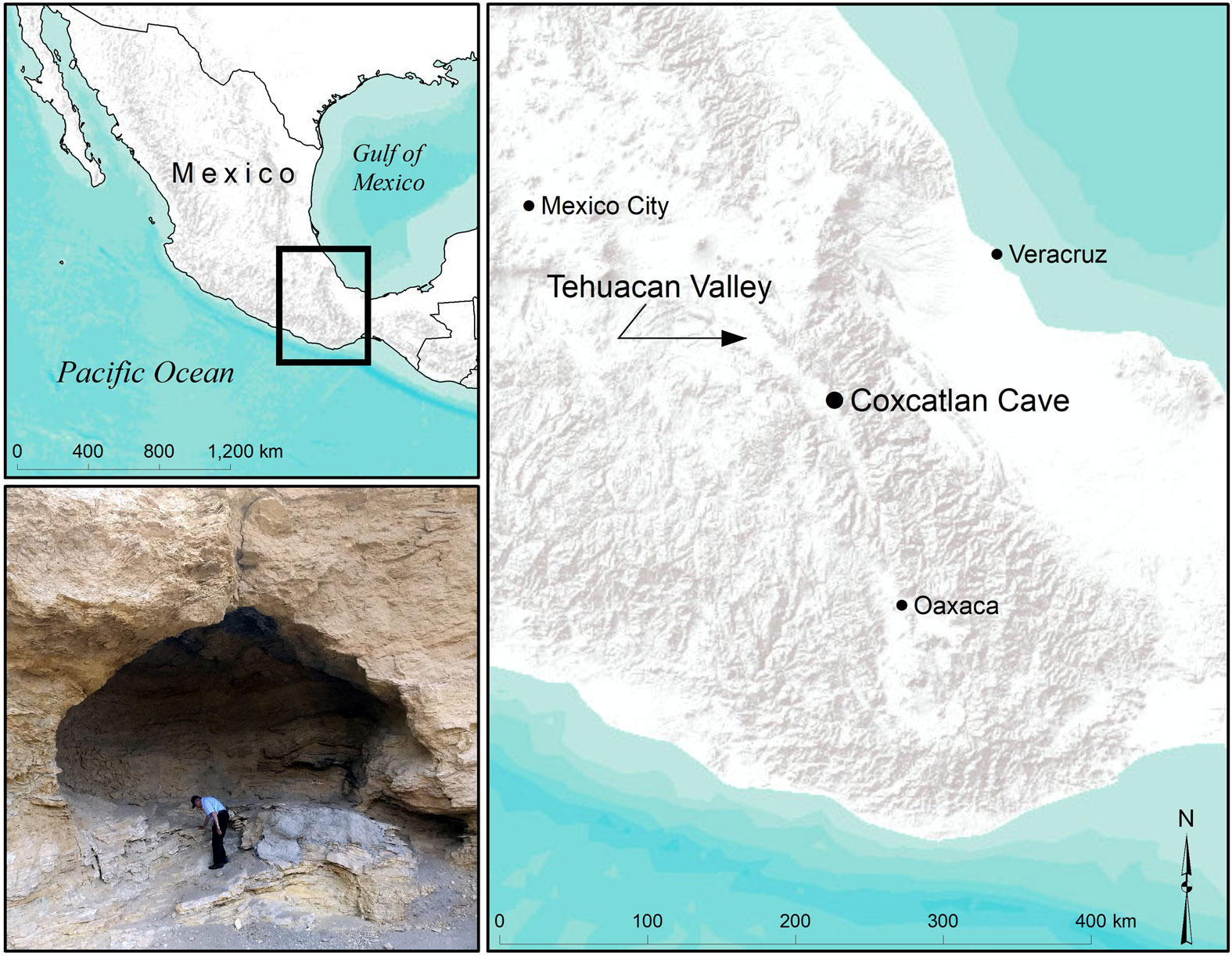
Maps showing the location of the Tehuacán Valley and Coxcatlán Cave in Mexico, as well as a picture of the cave (lower left). Photo by Andrew D. Somerville, Figure 1 from Somerville et al. (2021) Latin American Antiquity 32: 612-626 (Creative Commons Attribution 4.0 International license).
Ocampo Caves (Tamaulipas, Mexico)
Additional ancient maize cobs come from the Ocampo Caves in Tamaulipas, the oldest of which are about 4400 years old. Genetic analysis of cobs ranging from about 4400 to 2350 years old from the Ocampo Caves showed that they consistently had some alleles (gene variants) that are found in modern maize. These alleles affect the plant's growth form and the protein and starch in the kernels.
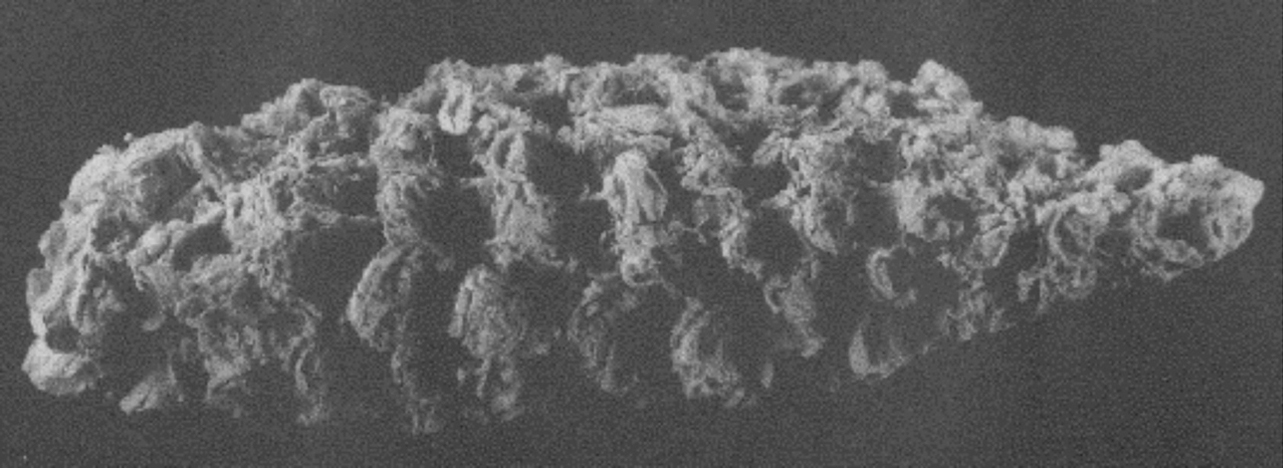
A Maize cob from Ocampo Caves, about 3890 years old, Tamaulipas, Mexico. The length of this cob is about 4.7 centimeters (1.85 inches). Photo by Svante Pääbo (Wikimedia Commons, Creative Commons Attribution 2.5 Generic license, image rotated).
El Gigante rockshelter (Honduras)
The El Gigante site in Honduras preserves well-dated maize cobs spanning from around 4300 years ago to less than 1200 years ago. The structures of the cobs at El Gigante show how maize ears changed through time as humans cultivated the crop over thousands of years. Most cobs at this site had 12 to 14 rows of kernels; a younger cob at the site had 22 rows of kernels.
The El Gigante cobs show that the history of maize domestication and improvement was complex. Genetic analysis of 2300- to 1900-year-old cobs from the site revealed that they are related to South American varieties of maize, both modern and ancient. Researchers think that maize first spread south from Mexico, and, later, that improved maize varieties spread back to the north from South America. The region of maize improvement in South America may have been in the southwestern Amazon.
Southwestern U.S.A.
Archeological sites with maize macroremains are also found in the Southwestern U.S.A. The oldest come from the Old Corn Site, New Mexico, and are about 4200 years old.
Researchers studied the genetics of maize samples from ancient sites in the Southwestern U.S.A. spanning 3390 to 670 years old. They compared the DNA from the ancient Southwestern maize with the DNA of Balsas and Mexican teosintes and other ancient and modern types of maize. The researchers concluded that while maize first reached the southwestern U.S. from the Mexican highlands, later genetic influences came from the Pacific coastal Mexican lowlands. Some of the younger maize had genetic similarities to a type of modern Mexican maize called Chapalote.

Maize of the Chapalote landrace. Photo by CIMMYT (flickr, Creative Commons Attribution-NonCommercial-ShareAlike 2.0 Generic license, image cropped and resized).
Maize becomes a staple crop
Analysis of human remains from Mesoamerica indicates that maize only became a significant part of some peoples' diets between about 4700 and 4000 years ago, shifting to a dietary staple after 4000 years ago. The rise in maize cultivation roughly coincided with the rise of more complex, densely populated societies that relied more heavily on agriculture in the Americas.
Increased cultivation of maize in particular did not necessarily precede or cause these social changes. Many other staple crops that could help sustain large populations were grown in the Americas, such as gourds, beans, cassava or manioc (Manihot esculenta, a tropical plant with a starchy root), and potatoes. Once Europeans arrived in the late 1400s, maize had become the widely grown, domesticated plant that we now recognize, with varieties suitable for cultivation in environments spanning from southern Canada far into South America.

Ancient ceramics depicting maize from Peru. Left: Vessel shaped like a maize plant, Nazca, 1 to 600 AD. Right: Vessel shaped like a rodent with an ear of maize, Moche, 50 to 800 AD. Left photo by the Metropolitan Museum of Art, right photo by the Cleveland Museum of Art, both photos CC0 1.0 Universal/Public Domain Dedication.

Figure of the Young Maize Deity of the Maya (an ancient Mexican civilization), ca. 900s AD, in three views. Left photo, center photo, and right photo by the Metropolitan Museum of Art (Wikimedia Commons, Creative Commons CC0 1.0 Universal Public Domain Dedication).

An ear of maize, 900 to 1275 AD, Sheep Horn Alcove, Utah. Photo by Daderot (Wikimedia Commons, Creative Commons CC0 1.0 Universal Public Domain Dedication).
Resources
Web resources
Origins of agriculture (Archaeological research in Oaxaca, Mexico, University of Michigan College of Literature, Science, and the Arts): https://sites.lsa.umich.edu/oaxaca-archaeology/origins-of-agriculture/
Books
Blake, M. 2015. Maize for the gods. University of California Press, Oakland, California.
Selected scientific papers
Benz, B. F. 2001. Archaeological evidence of teosinte domestication from Guilá Naquitz, Oaxaca. Proceedings of the National Academy of Sciences 98: 2104–2106. https://doi.org/10.1073/pnas.98.4.2104
Benz, B. F., and H. H. Iltis. 1990. Studies in archaeological maize I: The "wild" maize from San Marcos Cave reexamined. American Antiquity 55: 500-511.
da Fonseca, R. R., B. D. Smith, N. Wales, E. Cappellini, P. Skoglund, M Fumagalli, J. A. Samaniego, C. Carøe, M. C. Ávila-Arcos, D. E. Hufnagel, T. S. Korneliussen, F. G. Vieira, M. Jakobsson, B. Arriaza, E. Willersley, R. Nielsen, M. B. Hufford, A. Albrechtsen, J. Ross-Ibarra, and M. T. P. Gilbert. 2015. The origin and evolution of maize in the Southwestern United States. Nature Plants 1: 14003. https://doi.org/10.1038/nplants.2014.3
Grobman, A., D. Bonavia, T. D. Dillehay, D. R. Pipnero, J. Iriarte, and I. Holst. 2011. Preceramic maize from Paredones and Huaca Prieta, Peru. Proceedings of the National Academy of Sciences 109: 1755–1759. https://doi.org/10.1073/pnas.1120270109
Jaenicke-Després, V., E. S. Buckler, B. D. Smith, M. T. P. Gilbert, A. Cooper, J. Doebley, and S. Pääbo. 2003. Early allelic selection in maize as revealed by ancient DNA. Science 302: 1206-1208. https://doi.org/10.1126/science.1089056
Kennett, D. J., H. B. Thakar A. M. VanDerwarker, D. L. Webster, B. J. Culleton, T. K. Harper, L. Kistler, T. E. Scheffler, and K. Hirth. 2017. High-precision chronology for Central American maize diversification from El Gigante rockshelter, Honduras. Proceedings of the National Academy of Sciences 109: 9026–9031. https://doi.org/10.1073/pnas.1705052114
Kennett, D. J., K. M. Prufer, B. J. Culleton, R. J. George, M. Robinson, W. R. Trask, G. M. Buckley, E. Moes, E. J. Kate, T. K. Harper, L. O'Donnell, E. E. Ray, E. C. Hill, A. Alsgaard, C. Merriman, C. Meredith, H. J. H. Edgar, J. J. Awe, and S. M. Gutierrez. 2020. Early isotopic evidence for maize as a staple grain in the Americas. Science Advances 6: eaba3245. https://doi.org/10.1126/sciadv.aba3245
Kisler, L., S. Y. Maezumi, J. G. De Souza, N. A. S. Przelomska, F. M. Costa, O. Smith, H. Loiselle, J. Ramos-Madrigal, N. Wales, E. R. Ribeiro, R. R. Morrison, C. Grimaldo, A. P. Prous, B. Arriaza, M. T. P. Gilbert, F. De Oliveira Freitas, and R. G. Allaby. 2018. Multiproxy evidence highlights a complex evolutionary legacy of maize in South America. Science. 362: 1309–1313. https://doi.org/10.1126/science.aav0207
Kistler, L., H. B. Thakar, A. M. VanDerwarker, A. Domic, A. Bergström, R. J. George, T. K. Harper, R. G. Allaby, K. Hirth, and D. J. Kennett. 2020. Archaeological Central American maize genomes suggest ancient gene flow from South America. Proceedings of the National Academy of Sciences 117: 33124–33129. https://doi.org/10.1073/pnas.2015560117
Piperno, D. R., and K. V. Fannery. 2001. The earliest archaeological maize (Zea mays L.) from highland Mexico: New accelerator mass spectrometry dates and their implications. Proceedings of the National Academy of Sciences 98: 2101–2103. https://doi.org/10.1073/pnas.98.4.2101
Piperno, D. R., A. J. Ranere, I. Holst, J. Iriarte, R. Dickau. 2009. Starch grain and phytolith evidence for early ninth millennium B.P. maize from the Central Balsas River Valley, Mexico. Proceedings of the National Academy of Sciences 106: 5019–5024. https://doi.org/10.1073/pnas.0812525106
Ramos-Madrigal, J., B. D. Smith, J. V. Moreno-Mayar, S. Gopalakrishnan, J. Ross-Ibarra, M. T. P. Gilbert, and N. Wales. 2016. Genome sequence of a 5,310-year-old maize cob provides insights into the early stages fo maize domestication. Current Biology 26: P3195–3201. https://doi.org/10.1016/j.cub.2016.09.036
Smith, B. D 2017. Tracing the initial diffusion of maize in North America. Pp. 332–348 in N. Boivin, ed. Human dispersal and species movement. Cambridge University Press, Cambridge, U.K.
Stitzer, M. C., and J. Ross-Ibarra. 2018. Maize domestication and gene interaction. New Phytologist 220: 395–408. https://doi.org/10.1111/nph.15350
Vallebueno-Estrada, M., I. Rodríguez-Arévalo, A. Rougon-Cardoso, J. Martínez González, A. García Cook, R. Montiel, and J.-P. Vielle-Calzada. 2016. The earliest maize from San Marcos Tehuacán is a partial domesticate with genomic evidence of inbreeding. Proceedings of the National Academy of Sciences 113: 14151–14156. https://doi.org/10.1073/pnas.1609701113
Wang, H., A. J. Studer, Q. Zhao, R. Meeley, and J. F. Doebley. 2015. Evidence that the origin of naked kernels during maize domestication was caused by a single amino acid substitution in tga1. Genetics. 200: 965-974. https://doi.org/10.1534/genetics.115.175752
Webster, D. L. 2011. Backward bottlenecks. Current Anthropology 52: 77-104. https://doi.org/10.1086/658400



